GW-SOC81
This is a benchmark of G0W0 on 81 molecules, introduced in [1], for testing the implementation of G0W0 calculations including spin-orbit coupling (SOC). Each molecule has at least one heavy element (Z ≥ 29). For each molecule, the vertical ionization potential (VIP) is computed with the WEST code.
Benchmark summary
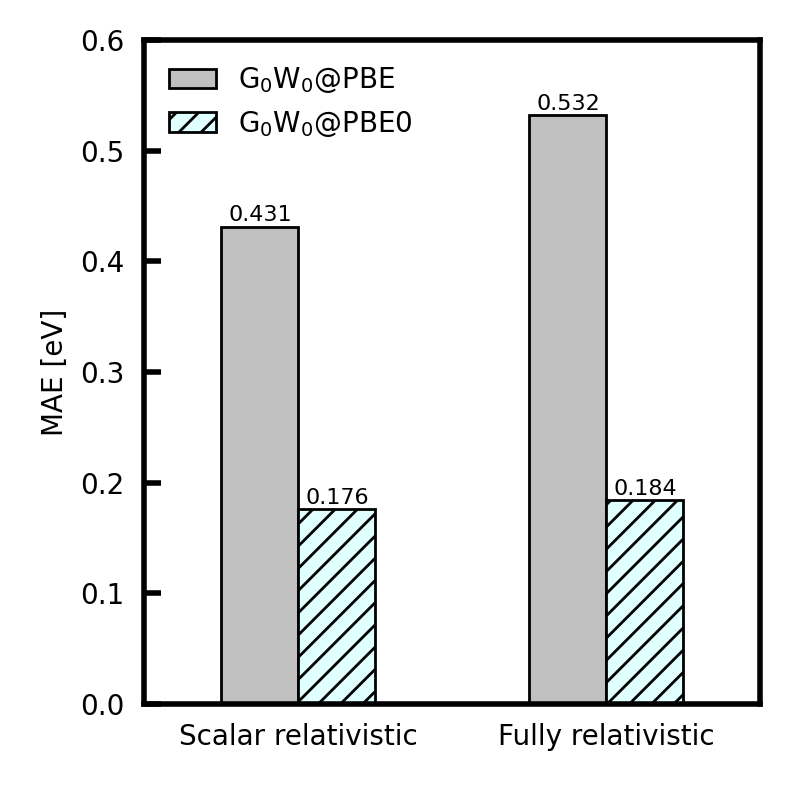
The figure below reports the mean absolute error (MAE) in the VIPs with respect to experiment. Values of WEST are taken from [1]. The VIP is computed from the quasiparticle energy of the highest occupied molecular orbital (HOMO), using scalar or fully relativistic pseudopotentials and the PBE or PBE0 exchange-correlation functional. Experimental results are taken from [2].
List of all molecules
Click the molecule name to see the dielectric screening, spectral function, and VIP computed with the WEST code.
| name | formula | CAS number | picture | |
|---|---|---|---|---|
| 1 | silver bromide | AgBr | 7785-23-1 |  |
| 2 | silver chloride | AgCl | 7783-90-6 |  |
| 3 | silver iodide | AgI | 7783-96-2 |  |
| 4 | dialuminum hexabromide | Al2Br6 | 18898-34-5 |  |
| 5 | aluminum bromide | AlBr3 | 7727-15-3 |  |
| 6 | aluminum iodide | AlI3 | 7784-23-8 |  |
| 7 | arsenic tribromide | AsBr3 | 7784-33-0 |  |
| 8 | arsenic trichloride | AsCl3 | 7784-34-1 |  |
| 9 | trifluoroarsine | AsF3 | 7784-35-2 |  |
| 10 | pentafluoroarsorane | AsF5 | 7784-36-3 |  |
| 11 | arsine | AsH3 | 7784-42-1 |  |
| 12 | arsenic triiodide | AsI3 | 7784-45-4 |  |
| 13 | bromine dimer | Br2 | 7726-95-6 |  |
| 14 | bromine monochloride | BrCl | 13863-41-7 |  |
| 15 | selenoketene | C2H2Se | 61134-37-0 |  |
| 16 | bromoketene | C2HBrO | 78957-22-9 | 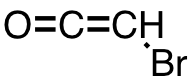 |
| 17 | selenophene | C4H4Se | 288-05-1 | 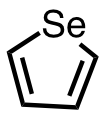 |
| 18 | bis(cyclopentadienyl)ruthenium(II) | (C5H5)2Ru | 1287-13-4 | 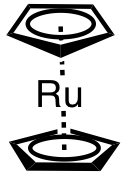 |
| 19 | trifluoroiodomethane | CF3 | 2314-97-8 | 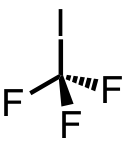 |
| 20 | dimethylcadmium | (CH3)2Cd | 506-82-1 | 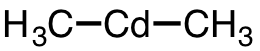 |
| 21 | dimethylmercury | (CH3)2Hg | 593-74-8 | 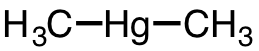 |
| 22 | dimethylselenide | (CH3)2Se | 593-79-3 | 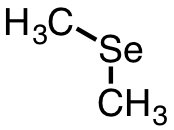 |
| 23 | dimethylzinc | (CH3)2Zn | 544-97-8 | 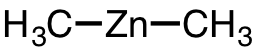 |
| 24 | bromomethylmercury | CH3 | 506-83-2 | 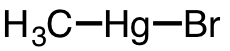 |
| 25 | chloromethylmercury | CH3 | 115-09-3 | 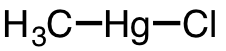 |
| 26 | iodomethylmercury | CH3 | 143-36-2 | 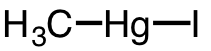 |
| 27 | iodomethane | CH3 | 74-88-4 | 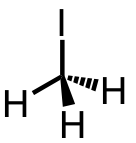 |
| 28 | tetraiodomethane | CI4 | 507-25-5 | 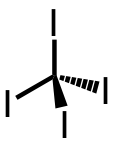 |
| 29 | calcium bromide | CaBr2 | 7789-41-5 |  |
| 30 | calcium iodide | CaI2 | 10102-68-8 | 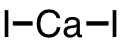 |
| 31 | cadmium bromide | CdBr2 | 7789-42-6 | 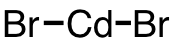 |
| 32 | cadmium chloride | CdCl2 | 10108-64-2 | 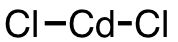 |
| 33 | cadmium iodide | CdI2 | 7790-80-9 |  |
| 34 | cesium chloride | CsCl | 7647-17-8 |  |
| 35 | cesium fluoride | CsF | 13400-13-0 |  |
| 36 | cesium iodide | CsI | 7789-17-5 |  |
| 37 | copper monofluoride | CuF | 13478-41-6 |  |
| 38 | mercury(II) chloride | HgCl2 | 7487-94-7 |  |
| 39 | iodine dimer | I2 | 7553-56-2 |  |
| 40 | iodine monobromide | IBr | 7789-33-5 |  |
| 41 | iodine monochloride | ICl | 7790-99-0 |  |
| 42 | iodine monofluoride | IF | 13873-84-2 |  |
| 43 | potassium bromide | KBr | 7758-02-3 |  |
| 44 | potassium iodide | KI | 7681-11-0 |  |
| 45 | krypton dimer | Kr2 | 12596-40-6 |  |
| 46 | krypton difluoride | KrF2 | 13773-81-4 |  |
| 47 | lanthanum tribromide | LaBr3 | 13536-79-3 |  |
| 48 | lanthanum trichloride | LaCl3 | 10099-58-8 |  |
| 49 | lithium bromide | LiBr | 7550-35-8 |  |
| 50 | lithium iodide | LiI | 10377-51-2 |  |
| 51 | magnesium bromide | MgBr2 | 7789-48-2 | 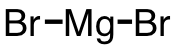 |
| 52 | magnesium iodide | MgI2 | 10377-58-9 | 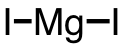 |
| 53 | molybdenum hexacarbonyl | Mo(CO)6 | 13939-06-5 | 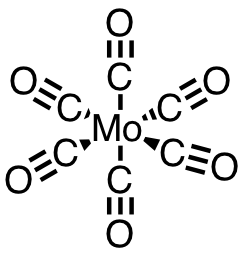 |
| 54 | sodium bromide | NaBr | 7647-15-6 | 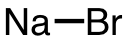 |
| 55 | sodium iodide | NaI | 7681-82-5 |  |
| 56 | osmium tetroxide | OsO4 | 20816-12-0 |  |
| 57 | phosphorus tribromide | PBr3 | 7789-60-8 |  |
| 58 | phosphoryl tribromide | POBr3 | 7789-59-5 |  |
| 59 | rubidium bromide | RbBr | 7789-39-1 | 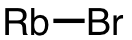 |
| 60 | rubidium chloride | RbCl | 7791-11-9 | 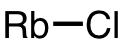 |
| 61 | rubidium iodide | RbI | 7790-29-6 | 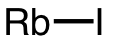 |
| 62 | ruthenium tetroxide | RuO4 | 20427-56-9 | 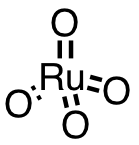 |
| 63 | thionyl dibromide | SOBr2 | 507-16-4 |  |
| 64 | thiophosphoryl tribromide | SPBr3 | 3931-89-3 |  |
| 65 | selenium dichloride | SeCl2 | 14457-70-6 |  |
| 66 | selenium(IV) oxide | SeO2 | 7446-08-4 |  |
| 67 | bromotrifluorosilane | SiBrF3 | 14049-39-9 |  |
| 68 | iodosilane | SiH3 | 13598-42-0 |  |
| 69 | strontium bromide | SrBr2 | 10476-81-0 |  |
| 70 | strontium chloride | SrCl2 | 10476-85-4 |  |
| 71 | strontium iodide | SrI2 | 10476-86-5 |  |
| 72 | strontium oxide | SrO | 1314-11-0 |  |
| 73 | titanium tetrabromide | TiBr4 | 7789-68-6 |  |
| 74 | titanium tetraiodide | TiI4 | 7720-83-4 |  |
| 75 | zinc bromide | ZnBr2 | 7699-45-8 |  |
| 76 | zinc chloride | ZnCl2 | 7646-85-7 |  |
| 77 | zinc fluoride | ZnF2 | 7783-49-5 |  |
| 78 | zinc iodide | ZnI2 | 10139-47-6 |  |
| 79 | zirconium tetrabromide | ZrBr4 | 13777-25-8 |  |
| 80 | zirconium tetrachloride | ZrCl4 | 10026-11-6 |  |
| 81 | zirconium tetraiodide | ZrI4 | 13986-26-0 |  |
Reference
- Implementation and Validation of Fully-Relativistic GW Calculations: Spin-Orbit Coupling in Molecules, Nanocrystals and Solids, P. Scherpelz, M. Govoni, I. Hamada, and G. Galli, J. Chem. Theory Comput. 12, 3523 (2016).
- P.J. Linstrom and W.G. Mallard, Eds., NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg MD, 20899.